5 Steps To Mastering Global Clinical Trial Disclosure Compliance
Executive Summary
Clinical trial disclosure compliance must include identification and documentation of registry requirements, key stakeholders, company policies, systems, and processes.
Many biopharma companies remain challenged to comply with mandatory government requirements for disclosing clinical trial information, whether due to lack of knowledge or lack of effective corporate policies and procedures. A strategy including documenting disclosure requirements and trials subject to disclosure, identifying internal stakeholders with compliance responsibilities, implementing policies explicitly endorsed by top management, and tapping into useful tech tools can help remedy the problem.
The stakes can be significant. With increasing scrutiny by transparency advocates and the start of inspections by health authorities, the current level of noncompliance is damaging to the industry’s reputation and potentially costly for individual sponsors. (Also see "The Hidden Implications of Clinical Trial Disclosure Noncompliance" - Pink Sheet, 18 Mar, 2021.) However, while global trial disclosure requirements can be complex, sponsors can implement a straightforward five-step process to take control of trial transparency.
(Article continues after graphic.)
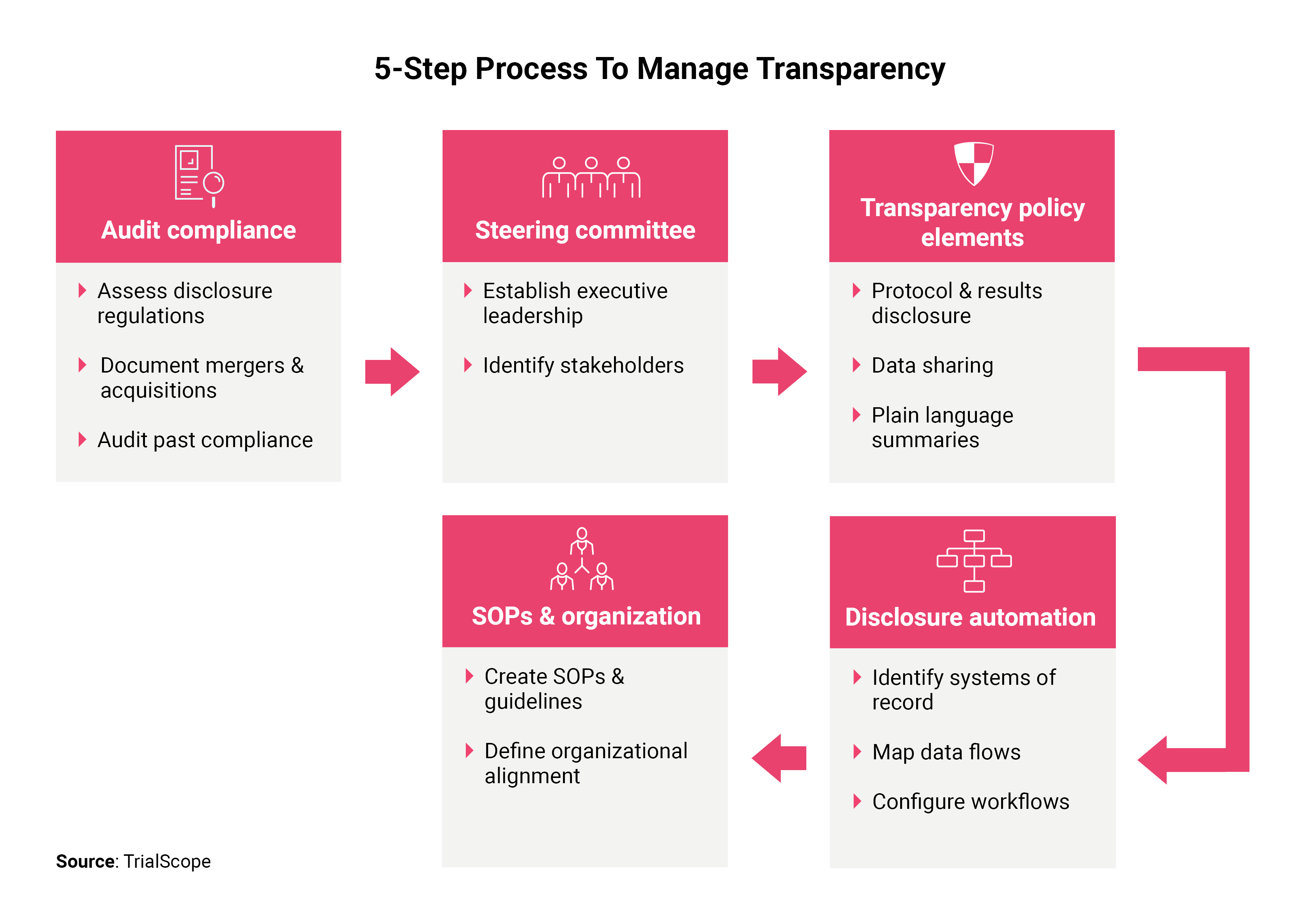
Key Steps Explained
Audit compliance and document regulations
Before developing policies or implementing a disclosure system, companies should document regulations and identify past compliance issues.
- Document acquisitions, mergers, and divestitures: To determine which trials to audit and which country’s disclosure regulations to assess first, document all acquisitions, mergers, name changes, and divestitures. The resulting list of products and business entities will be used to identify the trials for which the organization is responsible.
- Assess disclosure regulations: List the countries in which trials associated with the organization’s products and the business entities were conducted, going back at least 10 years, and forecasting anticipated site locations for the next 24 months. Determine the disclosure regulations for each country in which trials are conducted.
-
Audit past compliance: Assess disclosure compliance for the trials associated with the business entities and products identified above, comparing disclosure status for each trial with the regulations documented for the countries in which it was conducted. Since inspections have started in the US and EU, prioritize ClinicalTrials.gov and EudraCT, two significant registries that are frequently used for analysis by transparency advocates and industry critics. Compliance on every mandatory registry is equally important, but these are suggested as a starting point because the requirements are well known and the audit can be supported by two public compliance trackers, the FDAAA TrialsTracker and the EU Trials Tracker.
A Closer Look At Transparency
This is the third article in our series on clinical trial disclosure. Previous articles addressed the hidden costs of noncompliance and challenges faced by smaller companies.
Establish a transparency steering committee and identify stakeholders
It's wise to establish a transparency steering committee before creating a corporate transparency policy. This committee typically includes the chief medical officer and executives from clinical development and operations, and medical writing, and may include leaders from other departments such as patient engagement, corporate communications, and marketing. The primary purpose is to define the scope of clinical trial transparency, especially if these commitments go beyond the minimum required by law, and to communicate executive support for the transparency policies.
The public disclosure of clinical data requires the input of various departments and may affect the timing and communications of other public releases of the trial information. In addition to the department or individuals directly responsible for preparing the data and managing process, three key stakeholder groups are:
- Reviewers: Responsible for carefully reviewing the data prepared for disclosure to ensure that it is complete, accurate, and does not include personally identifiable or company confidential information, unless required by law. Reviewers are typically from clinical operations, regulatory affairs, biostatistics, and medical writing.
- Approvers: Responsible for approving the final versions of disclosure information prior to public release. Approvers are often the heads of clinical development or therapeutic areas.
- Informed; Stakeholders who should be informed prior to the public release of clinical data, including investor relations, corporate communications, legal (patent and regulatory), and publications leads. While these groups usually do not have reviewer or approver roles, they should be notified with enough time to prepare their response to the dissemination of the clinical data.
Create a transparency policy
To respond to disclosure regulations and expectations by patient groups and industry critics, organizations should develop or update their transparency policies. This document clarifies the organization’s commitments, shapes the disclosure standard operating procedures (SOPs), and establishes the organization’s culture around clinical data dissemination.
The policy’s level of detail will vary by company but should address the scope of commitments for protocol and results disclosure, data sharing, and plain language summaries. Typically, the policy states to what degree the company will make data available beyond the regulatory requirements and establishes timeframes for these commitments. [Editor’s note: See the Informa Pharma Intelligence Clinical Trial Policy Guide for a more detailed reference on developing an organization transparency policy.]
Implement disclosure system
Managing disclosure efficiently requires some level of automation. Specialized disclosure systems provide the greatest functionality, but home-grown solutions may work to support the disclosure team. Whatever system is implemented, it requires:
- Identifying the system of record for the data, which may be the CTMS, the protocol, SAS, the CSR, a TMF system, or data files sent by CROs and partners.
- Mapping data flows from the systems of record through data entry to the registries, as well as mapping the data relationships between registries to maximize data reuse and minimize discrepancies in the publicly available data.
- Configuring the workflows to assign disclosure, review, approval, and registry submission tasks
- Computer system validation, typically conforming to the US 21 CFR Part 11 and EU Annex 11 requirements.
SOPs & organizational alignment
SOPs define the consistent and repeatable disclosure processes to meet regulatory requirements and the commitments defined in transparency policy.
Organizations working with an external trial disclosure company are advised to finalize the SOPs and guidelines based on the workflows and capabilities of the contract partner. Postponing process documentation until later can impair custom configuration of services from the partner, missing out on efficient best practice workflows and raising the cost of implementation and testing.
Companies lacking a central coordinating function have the least assurance of compliant and consistent global disclosure.
(Article continues after graphic.)
 FDAAA TrialsTracker & TrialScope Assessment - February 2021
FDAAA TrialsTracker & TrialScope Assessment - February 2021
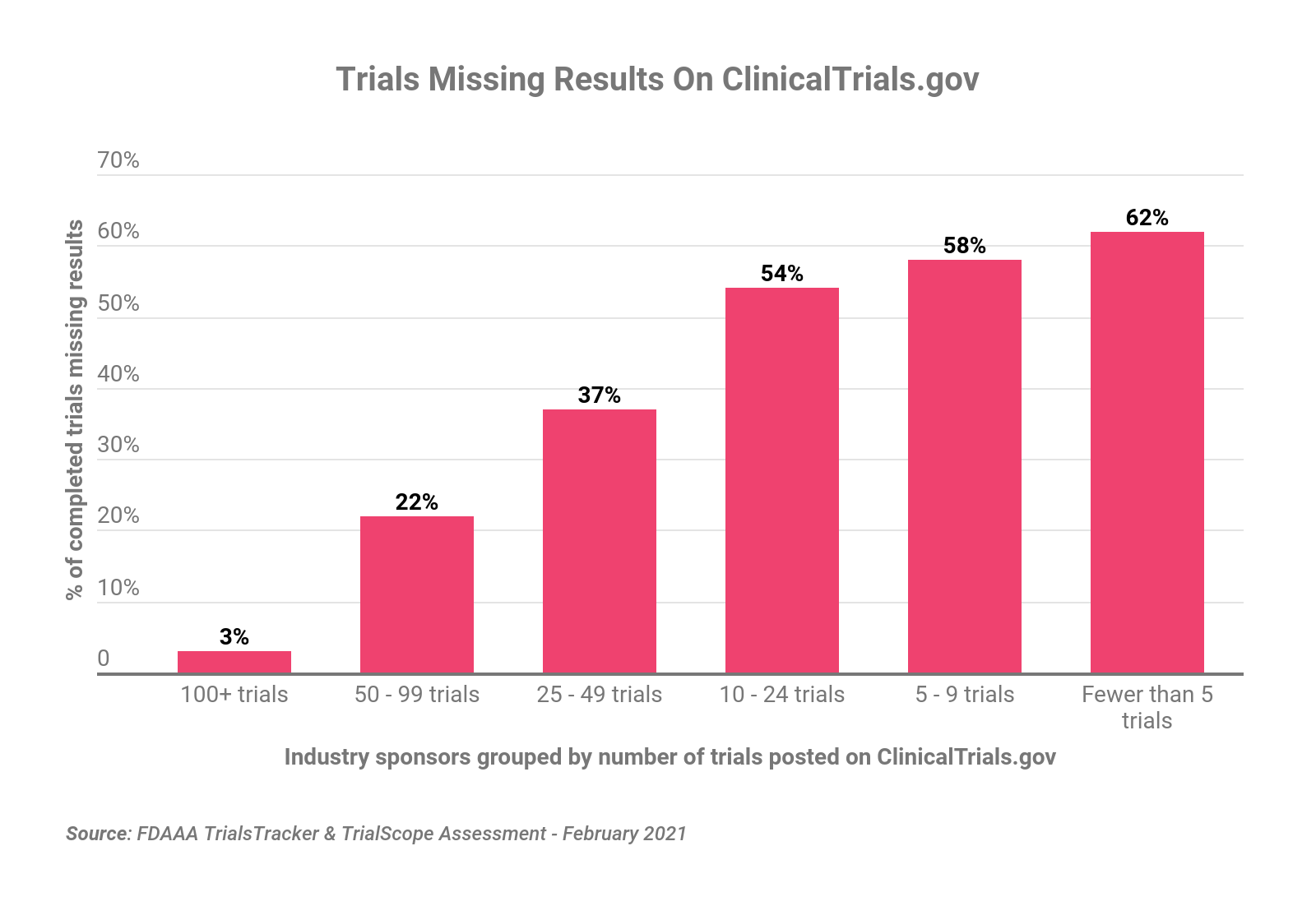
Sponsors with larger trial portfolios are recommended to establish a dedicated department, with responsibility for managing disclosure across all regions. The department should track the regulations for every country in which the company conducts clinical trials, prepare a standard set of data that can be disclosed to the public, and coordinate with affiliates and partners to ensure that only the data approved for disclosure are submitted to local registries. Mid-size companies often hire a disclosure expert responsible for submissions in the US and EU who coordinates with local affiliates or service providers for disclosure to the local registries. Smaller organizations should consider outsourcing the work to an experienced disclosure services provider managed by someone in regulatory affairs or regulatory writing.
The different challenges facing these different kinds of companies is evident in compliance rates. While the largest pharmaceutical companies have made significant strides in recent years, midsize and smaller companies are lagging behind considerably. For example, sponsors with 100-plus trials posted on ClinicalTrials.gov are more than 97% compliant in providing the required trial results records, while companies with fewer than 10 trials fall below a 40% compliance rate. (See chart above.)
Thomas Wicks is chief strategy officer at TrialScope. He has more than 20 years of experience with performance and content management approaches in the life sciences, with particular focus on clinical trial transparency.
.jpg?rev=302b1310147f4c219c8586e69fce802e&h=141&w=120&la=en&hash=A4CA4649E83F30DFDE0CB28209DEF1E5)