NIH Eyes Expansion Of US FDA Partnerships To Gene Therapy
Executive Summary
NIH Director Francis Collins describes evolution of existing collaborations and how the two agencies might facilitate development of gene therapy, such as by having a common template for regulatory review.
The National Institutes of Health is looking to build on its partnerships with the US Food and Drug Administration to tackle new challenges in gene therapy.
"We need to figure out every feasible way to facilitate" the development of gene therapy in a safe and effective way with FDA and industry, NIH Director Francis Collins said at an FDA Science Forum last month. He noted that he had a discussion with FDA's Peter Marks, director of the Center for Biologics Evaluation and Research, just two days earlier about how to move forward in this space.
One thing Marks is talking about is having a common template for regulatory review, Collins said.
Investigators would appreciate this, Collins said, as many of them will be working on rare diseases and may not have previously come before the agency to know how to get a successful review.
He noted several other things that need to be addressed, including having common platforms and improved vectors that are well studied and not tied up in intellectual property. “Manufacturing science needs to be improved as well because right now the ability to make high enough AV vector that you might want to use for a few patients is a pretty big challenge,” he said.
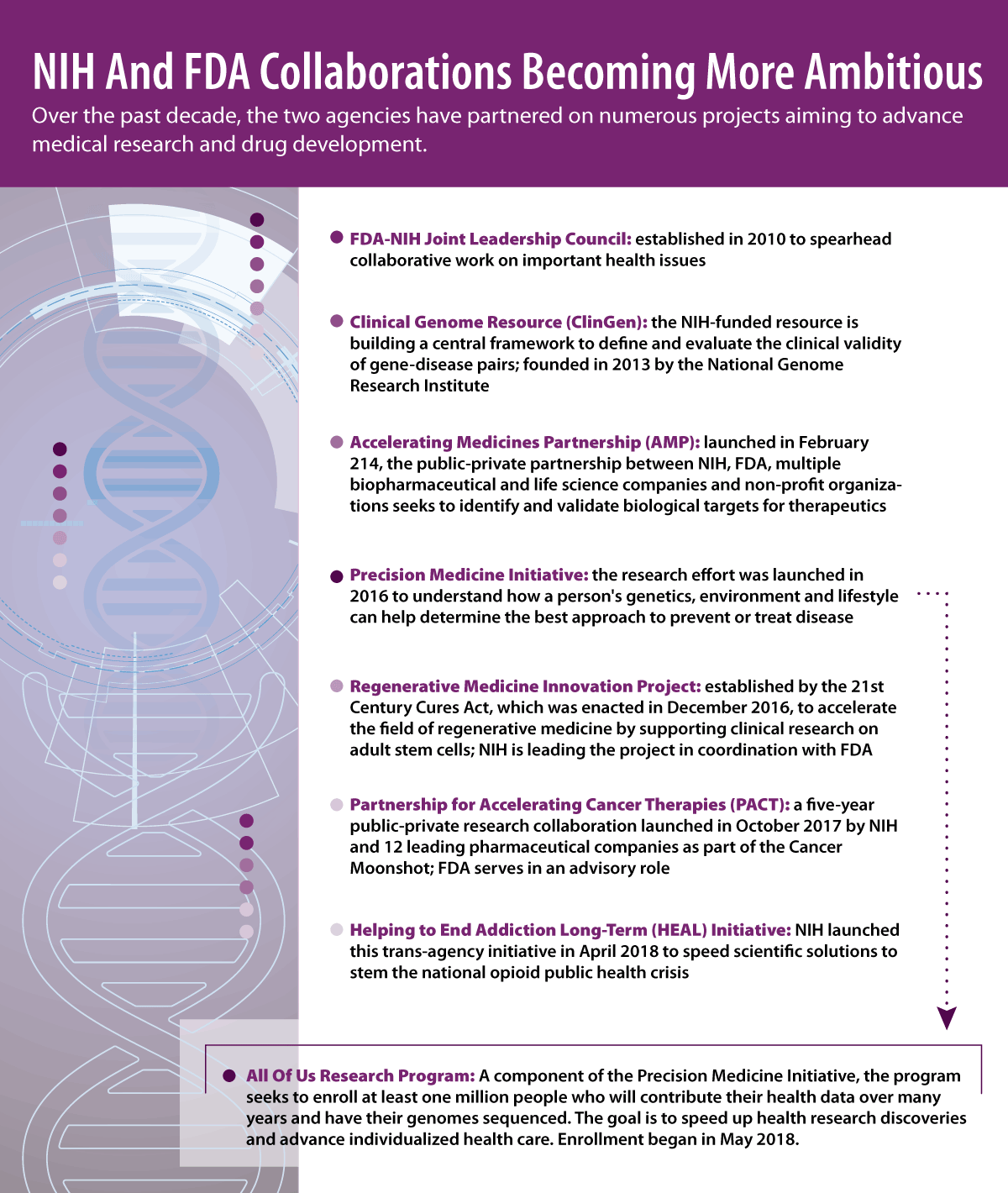
Collins focused on the partnerships NIH and FDA have had over the past decade to speed up the translation of research discoveries to interventions that reduce illness and disability. (See table below of key initiatives.)
Recombinant DNA Advisory Committee Is Reborn
The two agencies formed the FDA-NIH Joint Leadership Council in 2010, which Collins said was intended to make sure they did not just respond to things that came along in a reactive way but to work together proactively. One of the developments to come out of the council was the NIH-FDA Protocol Template for Phase II and Phase III investigational new drug/investigational device exemption clinical trials.
Collins noted that the current priorities of the leadership council include nutrition research, next generation sequencing, imaging and the opportunity to use machine learning on images and to fold this into radiomics.
The council is also focusing on modernizing human gene therapy. To that end, earlier this year NIH eliminated requirements for human gene transfer protocols to be submitted to NIH and reviewed by its Recombinant DNA Advisory Committee (RAC). (Also see "NIH Ends Gene Therapy Trial Reporting Requirements In Reg Streamlining Move" - Pink Sheet, 29 Apr, 2019.)
"Over the course of a year, with the help from the Institute of Medicine, now the National Academy of Sciences, we came to the conclusion that FDA's review process for gene therapy is now so well done, so rigorous, so effective in terms of assessing whether or not a particular therapy trial is appropriately designed, that we no longer needed to duplicate that" with RAC, he said. That duplication was "vexing to investigators who thought they had double jeopardy."
Collins said RAC has been reborn as NExTRAC (Novel and Exceptional Technology and Research Advisory Committee), which will have its first meeting in a couple of months.
The new committee "basically steps aside from the review of individual protocols and provides a public forum for debate about really truly significant new approaches that otherwise might not have a place to get discussed," he said.
Looking For Industry Assets For Phase II Chronic Pain Trials
Another collaboration Collins highlighted is the Accelerating Medicines Partnership, which was formed in 2014 with five years of funding commitments by NIH and industry to identify and validate targets for therapeutics. (Also see "NIH And 10 Biopharmas Hatch Plan To Accelerate And Improve Therapeutic Pathway Research" - Pink Sheet, 19 Feb, 2014.)
The public-private partnership, which now includes 12 life science companies, has projects in four disease areas: Alzheimer's disease, type 2 diabetes, autoimmune disorders of rheumatoid arthritis and lupus, and Parkinson's disease. Collins said the partnership is exploring a fifth project on schizophrenia.
"People were skeptical" about whether the partnership would work in bringing people together from different sectors, he said. But now all the projects are "in the process of debating how they might continue longer."
Collins also noted the progress the trans-agency Helping to End Addiction Long-Term (HEAL) Initiative has made since NIH launched it last year. The initiative is funding projects that seek to improve treatments for chronic pain, curb the rates of opioid abuse disorder and overdose, and achieve long-term recovery from opioid addiction. On 26 September NIH announced that through this initiative it has awarded $945m in total fiscal year 2019 funding for grants, contracts, and cooperative agreements across 41 states.
One of the areas HEAL is targeting is improving treatments for misuse and addiction. Collins said NIH has had discussions with FDA about ways the evaluation of such treatments might be broadened beyond the traditional consideration of whether individuals are able to achieve complete abstinence to other kinds of endpoints, such as reduction in craving.
NIH is also looking to industry to see what can be done with assets that companies have developed, but which currently are not on a fast track.
"We are setting up a clinical trials network to be populated with patients with chronic pain" in which Phase II trials can be done quickly, Collins said. "We have opened a website alternative opportunity for industry to volunteer assets that they would like to contribute to this. We have a whole system of how those will be prioritized and then put into the trial networks."
FDA Assessing How To Return Patients' Genetic Information
FDA also has a role in the biggest project to be undertaken by NIH, the All of Us Research Program. Launched in May 2018, the program aims to enroll at least one million people in the US, who will contribute their health data and have their genomes sequenced. The goal is to create a platform that all qualified researchers will have access to. As of the beginning of September, 300,000 participants had begun the program.
Collins said FDA is a critical part of the project as NIH is working with the agency to figure out what kind of health and genetic information can be released to participants who request it. He said NIH would propose that this include the ACMG 59, a set of 59 genes that the American College of Medical Genetics and Genomics has recommended be reported as actionable secondary findings in clinical exome and genome sequencing. Collins said most people agree you would want to know if you find a mutation in one of these genes that is actionable.
He said NIH also thinks people would be interested in pharmacogenomic data, particularly for drugs with a black box on the label specifying the importance of knowing your genotype before receiving a prescription.