China Hospital Drug Buying Slows, Biologics Outpace Market In 2013: McKinsey And CPA
This article was originally published in The Pink Sheet Daily
Executive Summary
Drug sales monitored at select hospitals in China rose at a double digit rate in 2013, but that is still a slower pace than in recent years as the impact of a compliance storm hit. Biologics however gained faster than small molecules and show potential for even greater growth, according to a recent report from global consulting firm McKinsey & Co and the Chinese Pharmaceutical Association.
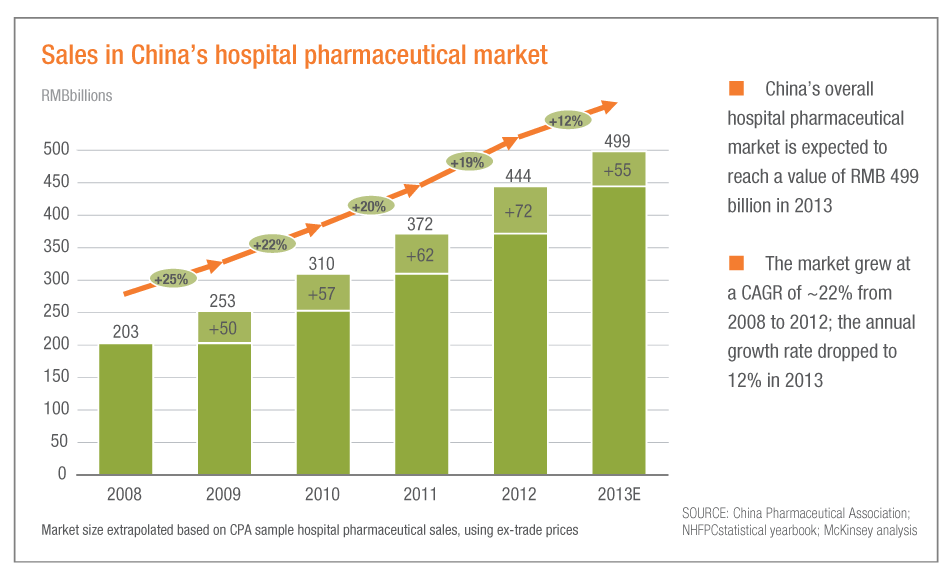
SHANGHAI – Drug sales at select Chinese hospitals slowed to a 12% annual pace in 2013 from a compound annual growth rate of 22% from 2008-2012, according to a recent China Hospital Pharmaceuticals Report released in February by the Chinese Pharmaceutical Association and McKinsey & Co.
In July last year the U.K. pharma giant GlaxoSmithKline PLC was investigated by Chinese authorities over its commercial practices (Also see "GSK Attempts to Stop The Rot And Maintain Market Share In China By Axing Employees" - Scrip, 8 Apr, 2014.). Then it spread to a national campaign on bribery violations in the pharma industry, which led to a slowdown of ex-manufacturer sales growth to 1% in the third quarter of 2013, based on figures reported by pharma companies (Also see "Behind The Ugly Numbers From The Compliance Crisis Fallout: China Big Pharma Roundup" - Scrip, 7 Nov, 2013.) and (Also see "Putting GSK’s China Bribery Crisis In Context" - Scrip, 18 Jul, 2013.).
However, the compliance fallout did not affect hospital purchasing volumes to the same extent as ex-manufacturer sales, said McKinsey, noting procurement at 650 sample hospitals across the country rose 11% in the third quarter of last year.
“The difference in growth rates for ex-manufacturer sales reported by companies and CPA hospital purchasing data likely occurred because distributors responded to the government’s measures by deferring orders and adopting a ‘wait-and-see’ strategy,” said McKinsey.
“The situation is still dynamic; we will need data for several more quarters before we can conclude if the market will return to historical growth rates or settle at a lower, but still double-digit, growth rate.”
Pharma markets in smaller cities still grew faster than first-tier cities. In 2013, tier 2 and tier 3 cities represented 58% of China’s hospital markets, in comparison, these mid-size cities account for 52% of the country’s GDP and 30% of the population, according to the report.
Multinationals have seen market share at tier 1 and tier 2 cities stable since 2010, which account for roughly half and one third market shares respectively.
Biologics Small, But Growing Faster
China’s biologics market is relatively small, accounting for 4.3% of the total prescription drug market, far behind the 21.5% share in the U.S. China also lags other emerging markets like Brazil, where biologics have a market share of 14.5%. Although the penetration gap shows the China biologics market is under-developed, it also indicates promising growth potential, said McKinsey.

The biologics market jumped 25% annually between 2010 and 2012, although it slowed to a 13% pace in 2013. Backed by the growing incidence of diseases like cancer and diabetes, economic development, favorable reimbursement policies and strong new product launches, the market is expected to continue its strong growth, said the report.
The report collected data from 508 big and middle size hospitals in 25 provinces, which account for approximately one quarter of the hospital market for biologics. Not surprisingly, Roche led with a 21% market share, flowing from its strong offerings of oncology products. The Swiss-based firm is followed by Denmark’s Novo Nordisk AS with its insulin products at 11% of the biologics market.
To be sure, the Chinese biologics market is more concentrated than small molecule offerings, with the top 10 players holding two thirds of the market at hospitals defined by McKinsey. Generally, multinationals dominate in the more innovative and more complex segments, and local champions claim the lion’s share in less complex molecules.
McKinsey’s Definition Of Biologics
- Biologic products include the following sub-segments:
Monoclonal antibody (mAb), fusion protein, recombinant coagulation factor, granulocyte colony stimulating factor (GCSF), growth factor, interleukin (IL), interferon (alpha, beta), insulin, erythropoietin (EPO), and hormone. The vaccine and plasma-extract segments are not included.
- These biologics are divided into complex molecules and less complex molecules:
The complex molecules include the mAb, fusion protein, and recombinant coagulation factor segments [accounts for roughly 30% of biologics market in the sample hospitals].
The less complex molecules include the GCSF, growth factor, IL, interferon, insulin (including analog), EPO, and hormone segments [accounts for roughly 70% of biologics market in the sample hospitals].
- The biologics market in sample hospitals represents ~60% of the complex biologics market and ~20% of less complex biologics market due to the different level of concentration in sample hospitals of different categories.
Top Brands For Complex Molecules
Mabthera (rituximab), Herceptin (trastuzumab) and Avastin (bevacizumab) together accounted for 54% of complex molecule sales in the third quarter of last year, making them the top three in sales of complex molecules.
Local brand Yisaipu (recombinant human TNF-A receptor II) from Shanghai CP Guojian Pharmaceutical Co. Ltd was at fourth place, thanks to a launch in 2005 ahead of major competitors in tumor necrosis factor inhibitors such as Johnson & Johnson’s Remicade (infliximab) [launched in 2006] Pfizer Inc.’s Enbrel (etanercept) [launched in 2010], andAbbVie Inc.’s Humira (adalimumab) [launched in 2011].
Another notable brand is Novartis AG’s Lucentis (ranibizumab), which was approved in 2011, and launched in China in 2012 (Also see "After Januvia And Onglyza, Novartis Launches Galvus In China; Lucentis Receives SFDA Nod" - Scrip, 19 Jan, 2012.). The drug has climbed to seventh place. Its quick success is largely built on the less competitive landscape, as it is the first licensed therapy in its class for wet age-related macular degeneration (AMD).
However, Chengdu Kanghong Pharmaceuticals (Group) Co. Ltd. launched Lang Mu (conbercept) at the end of last year, which “might pose a future challenge” to Lucentis, according to McKinsey.
There is huge potential in the oncology biologics market, said McKinsey, as Herceptin is the only available biologics brand from a multinational for the three most common cancers in China: lung, stomach and liver.
McKinsey cited the success of small molecules from multinationals such as Glivec/Gleevec (imatinib), Tarceva (erlotinib), Iressa (getfitinib) and said that could be a nugget for companies to explore.
The impressive market performance of Mabthera for lymphoma, the 12th most common cancer in China, “is one sign that there are major unmet needs in the market. If companies develop advanced treatments for more common cancers, they will tap into significant opportunities,” predicted McKinsey.
[Editor’s note: This story was contributed by PharmAsia News![]() , which provides in-depth coverage of Asia business and regulatory developments.]
, which provides in-depth coverage of Asia business and regulatory developments.]