Cancer’s Next-Gen Smart Bomb: Who Will Be First To Weaponize?
Executive Summary
With 70 clinical trials now underway on a “next generation” of more precisely targeted antibody drug conjugates, In Vivo profiles Ambrx and Sutro Biopharma, two smaller biotechs with promising technologies and powerful partners that augment their strong science with disease awareness, deep commercial networks and global geographic reach.
- Research is intensifying around the complex technology of antibody drug conjugates as a weapon against cancer.
- To date, ADCs have had only scattershot success in delivering cytotoxic “warheads” that bind to tumor cells and kill them, without the harsh patient side-effects that impair standard chemotherapy.
- Big pharma is active in the space, but some of the most significant work is being done by smaller biotechs such as Ambrx and Sutro.
- So what? Advances in ADCs, if realized in clinical trials, will provide patients with one of the first tangible benefits of precision medicine. ADCs may also answer the question that has dogged cancer treatment for decades: how to destroy a tumor without killing the patient in the process.
While innovations in drug discovery occupy center stage in the battle against cancer, it is the obscure mechanics of drug delivery that may have the edge in attacking – selectively, with laser-like precision – the mutant cell growth that leads to lethal tumors. New platform technologies linked to improved understanding of the genetic origins of most cancers are driving the creation of engineered protein antibodies that can be weaponized with toxins to single out cancer cells and kill them. The precise targeting of cancerous cells carries significant benefit to patients, minimizing the dangerous side effects of scorched earth chemotherapies, controlling the collateral damage to healthy tissue and raising the overall tolerability of a more potent treatment regimen.
The concept of using antibodies to fight cancer cell proliferation has been around for more than a decade, with two products currently in commercial use: Seattle Genetics Inc.'s Adcentris (brentuximab vedotin), approved by the FDA in 2011 for treatment of Hodgkin’s lymphoma; and the Roche drug Kadcyla (trastuzumab emtansine), FDA approved in 2013 for metastatic breast cancer. The basic thrust is the assembly of an antibody-based conjugate (ADC) drug consisting of an engineered cell antibody to seek out and bind to a tumor antigen cell, then release a cytotoxin “warhead” that kills it.
Interest in this field is strong mainly because the concept – and the emerging science behind it – promises to upend the scattershot response of conventional drug therapy against the relentless proliferation of mutant cells that cause cancer. According to Informa Pharma Intelligence’s Medtrack, three of the top 10 partnering deals in 2016 focused on the antibody space: a $3 billion deal between Merus NV and Incyte Corp. for a bispecific antibody platform for cancer [See Deal]; Celgene Corp.’s $2.5 billion deal with Jounce Therapeutics Inc. to access JTX-2011, an mAb investigational compound for cancer [See Deal]; and Novartis AG's acquisition from Xencor Inc. of another antibody platform, again, for cancer, at $2.6 billion. [See Deal]
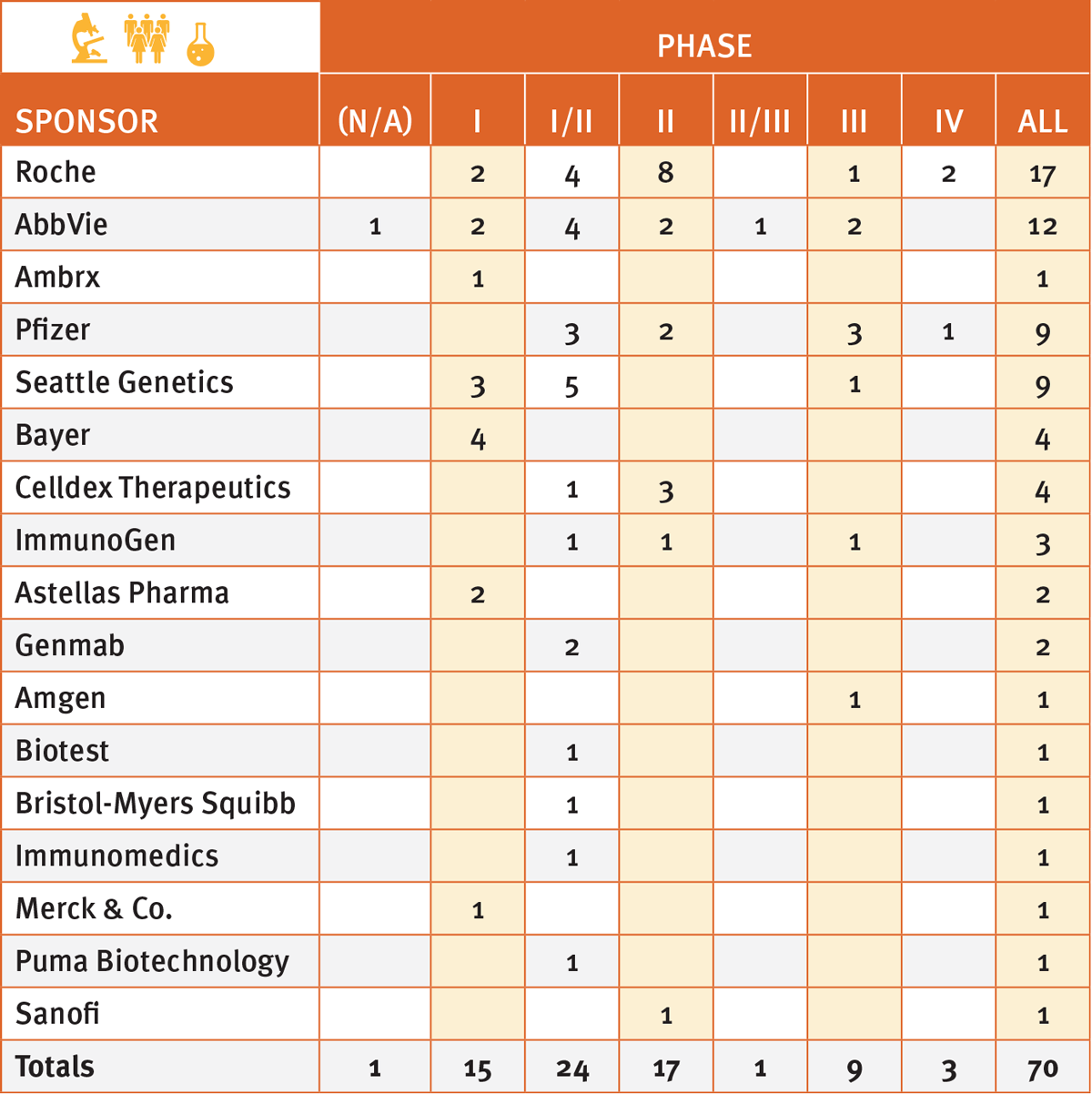
More important, some70 ADC compounds are presently in clinical trials, according to Pharma Intelligence's Trialtrove, mostly in the early test phases and involving six of the big pharma top 10, including Roche, the early leader in the field. (See Exhibit 1).
Exhibit 1
ADC Compounds In Clinical Trials
Trialtrove | Pharma Intelligence 2017
Pint-Size Potential
However, some of the most significant work on ADCs is underway in the smaller biotech segment. While Roche and Seattle Genetics are seeking to build on their first-to-market breakthroughs, two small California-based biotechs – privately held Sutro Biopharma Inc., founded in 2004, and Ambrx Inc., established in 2003 and now owned by a consortium of leading Chinese pharma players – are pursuing a variant path to enhance the effectiveness of ADC therapy in the individual patient.
Their approach centers on creating a stable, chemically homogenous and site-specific antibody warhead that dramatically increases the success rate in delivering that cytotoxic “payload” to the tumor cell target, precisely and at the fullest concentration that can be safely tolerated by the patient. It’s a thesis that, if realized in human trials, will refine and extend the clinical impact on tumor regression, in comparison with the current standard of care.
Although the technology differs, both companies appear evenly matched in pipeline potential. Ambrx’s lead ADC, ARX788, targeting the HER2 gene mutation in breast cancer, is now in Phase I trials in Australia and New Zealand. It also has an approved IND from the FDA for a similar trial in the US, to start later this year, as well as a novel off-licensing deal with Chinese partner Zhejiang Medicine Co. Ltd. (ZMC) through which the latter will coordinate a Chinese Phase I trial on ARX788, simultaneously with the one in the US. (Also see "Ambrx breaks deal mold with Zhejiang ADC alliance" - Scrip, 18 Jun, 2013.)
“We will initiate the Phase I US trial in September or October, involving a cohort of 50 patients for a duration of approximately one year,” Yong Hei, MD, chief medical officer for Ambrx, told In Vivo. “Depending on the results, we should be ready to commence to a Phase II trial by the end of 2018.” Hei also noted that the two trials in Australia and New Zealand on ARX788 are progressing well. “We’re optimistic that data from the trials will be available by mid-year 2018, perhaps along with some preliminary feedback on the US trial.”
Sutro has no candidate in trials as yet, but it expects to obtain INDs from the FDA for two ADC products within the next 12 to 18 months. First is STRO-001, an ADC that targets the protein CD74 associated with B-cell malignancies that cause non-Hodgkin's lymphoma and multiple myeloma. The second, STRO-002, targets the overexpression of the folate receptor alpha protein found in ovarian cancer and other solid tumors. Last month, at the annual meeting of the American Association for Cancer Research, Sutro announced results of an animal study on STRO-001 demonstrating potent anti-tumor activity against multiple myeloma, diffuse large B-cell lymphoma and mantle cell lymphoma models while reducing the potential for toxic secondary effects on adjacent healthy cells – a nearly ideal outcome for an ADC, albeit in mice.
In anticipation of future market commercialization, Sutro in March 2016 recruited two industry veterans to progress the company’s late-stage efforts on ADCs: former Johnson & Johnson oncologist Arturo Molina, MD, as chief medical officer; and Mark Lupher, PhD, in a new position as VP for translational pharmacology and preclinical development. In addition, Joseph Lobacki, former chief commercial officer of Medivation as well as lead manager for Genzyme’s global hematology business, was elected to Sutro’s board of directors last month.
Beyond these new leadership hires, Sutro has an exclusive claim to operating the world’s only cell-free cGMP manufacturing facility that eliminates reliance on complex, hard to replicate cultures of living cells in building an ADC arsenal. The patented process relies on transcripted material drawn from Escherichia coli bacteria to accelerate the speed and efficiency of a cell-free extract and protein base used to build an ADC; the time cycle for this process can be as short as 12 hours.
Big Pharma Connections
In addition, both companies can call on strong partnering links that carry differentiating advantages in geographic market reach. Sutro’s long-standing ties to Celgene, which began in 2012 and expanded into a formal strategic collaboration agreement in October 2014, gives it access to one of the industry’s strongest oncology sales and marketing networks. [See Deal]The two companies are currently advancing seven out of a total of 15 research and preclinical programs on multi-specific antibodies and ADCs. Sutro is responsible for early-stage research and preclinical development activities as well as the manufacturing of preclinical product candidates, whereas Celgene is responsible for global commercialization and, in that regard, holds or can acquire worldwide rights to market all products that stem directly from the collaboration.
The 2014 agreement burnishes the biotech’s bona fides by including an option for Celgene to acquire Sutro – that right expires in September but is subject to renewal through March 2019 if Celgene decides by September to request it. If Celgene opts not to acquire the company by the end of this period, US commercial rights for an unspecified number of the collaboration’s projects will revert to Sutro. It’s no surprise that this is when the evidence will be in hand as to the clinical potential of the STRO-001 and -002 ADC candidates.
Likewise, Ambrx, with its new owners, is poised to achieve a “world class standard” for ADC products in China, across a range of therapies. These owners, which include the Shanghai Fosun Pharmaceutical Group Co. Ltd.and WuXi PharmaTech Inc., carry marquee status in navigating China’s complex, politicized regulatory environment. [See Deal]With additional field-based support from ZMC, Ambrx’s Chinese partner on ARX788, these ties give company management better insights on how to build an ADC franchise not only in the world’s second largest drug market, but in other emerging middle-income countries as well. Supply chain logistics and manufacturing expertise are just a few of the capabilities these Chinese partners bring to the table. (Also see "Ambrx Looks Across Pacific For A Secure Future" - Scrip, 26 May, 2015.)
“China lags in medicines innovation and knows that currently the world has only two ADC anti-cancer products available, at a time when the technology behind this novel precision medicine platform is rapidly improving,” Ambrx CMO Hei said. “The Chinese regulatory authorities have been constructive in approving trials on ADC products – Roche already has a China trial underway on Kadcyla, its first-generation ADC product. Our partnership with ZMC on ARX788 complements these efforts, where we can see ARX788 being positioned in China not as a third-in-line treatment, but as potentially first-in-line.”
Technology Boost For Target Delivery
Kadcyla and Adcentris have had some success therapeutically with patients. Nevertheless, creating an ADC with a payload for accurate delivery to the tumor is complex, time-consuming and cumbersome against the infinite variations in tumor cell patterns – there is a “hit or miss” aspect to these first-generation technologies. Reports indicate that under current therapy, only about 1% of a systemic cytotoxic dose actually penetrates the tumor cell – obviously, that’s a number that bears improving.
And there is the need to develop living cell cultures to construct the ADC, which can take weeks or months, as well as to keep the constituent parts of the antibody stable in the bloodstream, ready to link seamlessly once they bind to the protein target on the surface of the tumor cell and insert the cytotoxin. This requires enormous precision in customizing an antibody capable of linking to the site, recognizing the tumor antigen, binding to it and ensuring the cytotoxin has sufficient potency to penetrate tumor cell defenses.
Drug design has thus centered on loading the antibody warhead with the molecular equivalent of multiple shots on target, using an assortment of different ADC molecules to maximize tumor kill potential. Unfortunately, that conventional approach has led to persistent drug efficacy issues like breakdown or partial impairment of the cytotoxin prior to reaching the tumor cell. This leaves the cancerous mutation unscathed along with dangerous side effects in patients when the cytotoxin is expressed not at the tumor site, but in the bloodstream or in otherwise healthy tissue. And the amount of cytotoxin delivered may be subpar, enabling tumor cells to develop resistance. The point is when the dose does not find its target, the desired therapeutic result – cancer regression at lower patient exposure to toxicities – is not achieved.
Do The Biotechs Have The Edge?
The good news is the science behind ADC is progressing. Rival biotechs Sutro and Ambrx contend their “third generation” technologies promise to boost the reliability of target delivery, providing a better patient experience and a superior clinical outcome. Here’s how they do it.
Sutro’s innovative step is twofold. First, it has introduced an artificial cell generation production technology that can be used interchangeably with different DNA strands to speed the assembly of ADCs, at lower cost, rather than replicating living cells on a one-time basis. Second, by enhancing the accuracy and stability of an ADC in delivering its cytotoxin payload directly to the tumor cell target, it improves the prospect for a safer and more clinically efficacious outcome for patients.
In a recent interview with In Vivo, Sutro CEO William Newell said, “For years, we’ve advocated that homogenous ADCs based on site-specific conjugation will improve efficacy and reduce toxicity to cancer patients. Many pharmaceutical companies have now come to adopt this view and they use different technologies to attempt to achieve it. What we’ve proven preclinically is that this is necessary but not sufficient to maximize the therapeutic index – the ratio between efficacy and toxicity – for an ADC. To do that, you have to select the optimal site (or sites) for attachment of a preferred linker and the cytotoxin. We believe Sutro’s Xpress CF+ platform technology is uniquely suited to do this, as we can access any site in an antibody as a place for the payload attachment – and we can do it all in only a few weeks, much faster than the norm with conventional discovery approaches. We can thus determine, by direct observation of a patient’s tumor profile, which site or sites are optimal. No one else in the industry can do that.” (See sidebar, "Q&A With Sutro's Bill Newell.)
Ambrx is pursuing the same end as Sutro, but with a structurally different approach focused on a process that replaces the amino acid in the cell protein with a non-natural variant of the amino acid. This facilitates the site-specific conjugation of a more concentrated toxin to attach to the tumor cell and kill it. “The Ambrx approach is radically homogenous,” said Chief Medical Officer Hei. “We have devised a technology hook that produces the necessary chemical reaction with the toxin without diversion into non-cancerous cell groups, translating into a better efficacy and safety profile for the patient.”
Hei also noted that the first-generation technology used by Roche and Seattle Genetics relies on a mixture of cytotoxins – as many as six – that tend to dilute the potency of the overall dose. “To us, homogenous is a synonym for concentrated; our model requires no more than two cytotoxins per warhead. And, in contrast to Sutro, our antibody is a natural antibody, generated from living cells. We think this promotes greater efficacy and tolerability for use in the human population.”
A Look Forward
There are two subtexts to these confident assessments of the therapeutic potential of next-generation ADCs. The first is that advancing targeted drug delivery while fighting the endless adaptability of a cancerous cell remains a daunting – some might say quixotic – task. Given the overall poor tumor penetration rates from the two ADCs in current use, any improvement must progress from a modest starting point. Literally, there is no place to go but up. How far and how fast pose contrasting implications for the future health of cancer patients, ranging from life-threatening side effects to full tumor regression.
The second is that the ADC space is competitive and crowded, with nearly 20 different platforms to deliver that lethal warhead in various stages of testing. All told, more than 40 pharma and biotech companies are engaged in next-generation ADC development, along with a variety of public and academic institutions, including the Scripps Research Institute, the National Cancer Institute, the University of Georgia, the University of California, Davis and University College London. “ADCs have clearly attracted the attention of Wall Street,” Les Fundleyder, portfolio manager for E Squared Capital Management, told In Vivo. “We are seeing an increase in transactions, which does suggest some third-party validation of the potential of the clinical class presently under development.”
As is the case in cancer treatment generally, scrutiny among payers on pricing is growing, so it is to be expected that new ADC regimens now in development will face market access issues based on comparisons against the current state of practice. A clear, clinically measurable advantage must be demonstrated around important oncology indicators like progression-free survival, backed up by evidence. In this regard, the therapeutic index – balancing efficacy with tolerability – will be determinative in establishing an appropriate P&R rate.
There are opportunities from the new science as well. These include the application of ADCs to other conditions beyond cancer, terrain that has been only lightly explored by big pharma to date. There is also promise in replacing or combining the toxic chemotherapy drugs as ADC “weapons of choice” with the checkpoint inhibitor immunotherapies that, in active clinical use, have brought virtual cures to some cancer patients. This would deliver a superior dose to combat the cancer while minimizing adverse effects on the patient. Likewise, new weapons in the ADC arsenal will discourage the onset of tumor resistance, the Achilles' heel in all cancer drug therapies.